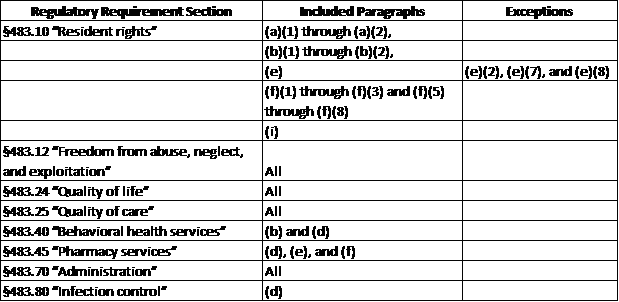
In a May 12, 2017 S&C letter addressed to nursing homes, The Centers for Medicare & Medicaid Services (CMS) provide an updated definition on Substandard Quality of Care (SQC) and how it will be implemented by Phase in the updated Requirements of Participation. In the updated RoPs, the definition for SQC was included without substantive changes, but the regulatory citations to the relevant requirements were updated. This means that until CMS updates the list of F-tag numbers, some of the information under certain tags will be included or excluded. The new definition of Substandard Quality of Care is:
“One or more deficiencies which constitute either immediate jeopardy to resident health or safety; a pattern of or widespread actual harm that is not immediate jeopardy; or a widespread potential for more than minimal harm, but less than immediate jeopardy, with no actual harm, related to participation requirements under . . .”
For each Phase of the RoPs, as mentioned above, SQC is implemented in the following ways:
- Phase 1: Most of the F-tags that were identified as SQC prior to November 28, 2016 are considered to be SQC. Those tags include the ones within these ranges:
- F221-F226
- F240-F258
- F309-F334
Simple enough, right? No. There are five “notable” exceptions that include F-tags that are now considered to be SQC or may now be considered SQC because only a portion of the regulation within the F-tag is defined as SQC. This leaves it up to the surveyor to determine which regulation they are citing. CMS notes that neither ASPEN nor the QIS software will be updated to identify these tags in Phase 1.
- Phase 1 F-tag exceptions:
- F151- Contains §483.10(b)(1) through (b)(2)
- F155 – Only consider SQC if the deficient practice is found at §483.24(a)(3)
- F175 – Contains §483.10(e)(4) and (5)
- F252 – Only consider SQC if the deficient practice is found at §483.10(i) and 483.10(i)(1)(i)-(ii)
- F461 – Only consider SQC if the deficient practice is found at §483.10(i)(4) or 483.25(n)(4)
In Phase 2, the new F-tag numbering system will be in place. This means that the affected F-tags will be spread out to their new regulatory groups and there will no longer be both SQC and non-SQC under the same groups.
- ASPEN and the new hybrid survey process software will be updated to identify the correct tags.
- Surveyors will be responsible for determining if the regulation that they are citing is SQC.
By Phase 3, all the issues will have been addressed and CMS does not anticipate any additional changes needed related to SQC.
Read CMS S&C memo “Implementation Issues, Long-Term Care Regulatory Changes: Substandard Quality of Care (SQC) and Clarification of Notice before Transfer or Discharge Requirements” (Ref: S&C: 17-27-NH) on the CMS website.